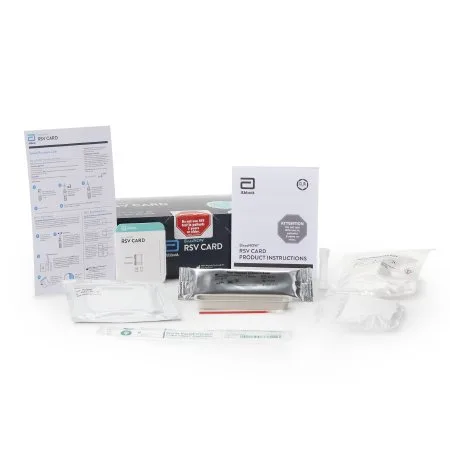
Alere BinaxNOW® RSV Card is an in vitro rapid immunochromatographic assay used to detect respiratory syncytial virus (RSV) fusion protein antigen in nasal wash and nasopharyngeal (NP) swab specimens from symptomatic patients
This test is intended for in vitro diagnostic use to aid in the diagnosis of respiratory syncytial virus infections in neonatal and pediatric patients under the age of 5
Retrospective nasal wash Sensitivity: 89% Specificity: 100%
Prospective NP swab Sensitivity: 93% Specificity 93%
Lab-Respiratory Testing, Rsv - Visually Read
TEST KIT, WAIVED BINAX NOW RSV22 (22/KT) WAMPL
Respiratory Test Kit BinaxNOW® Infectious Disease Immunoassay Respiratory Syncytial Virus Test (RSV) Nasopharyngeal Swab / Nasal Wash Sample 22 Tests CLIA Waived
| Units of Measurement | |
|---|---|
| Manufacturer | ABBOTT RAPID DX |
| UNSPSC | 41116144 |
| Product Specification Sheet | |
| SDS | SDS_WAMPL_TEST_KIT_WAIVED_BINAX_NOW_RSV221.pdf |
| BinaxNow RSV Card Product Instructions | BinaxNow_RSV_Card_Product_Instructions.pdf |